Predictive Pre-operative Planning for Medical Devices
Our foundation is built on a deep pedigree of research published in leading medical and engineering journals. We bridge the gap between engineering and medicine to solve the most complex challenges in structural heart disease through advanced constitutive modeling, patient-specific simulation, and structural biomechanics.
Transcatheter Aortic Valve Replacement (TAVR)
Patient-specific biomechanical simulation for aortic valve procedures
Virtual Valve Deployment & Rupture Risk
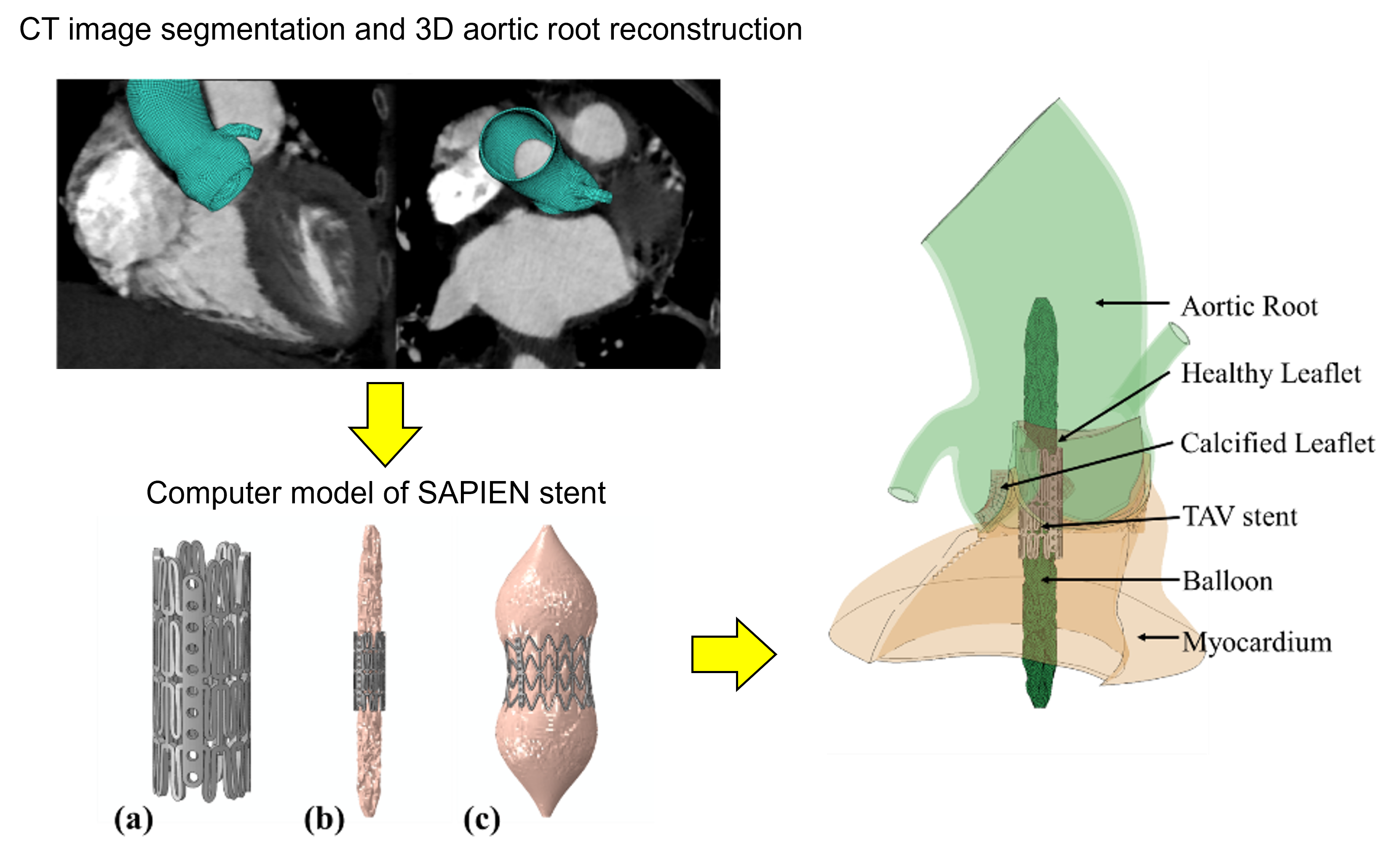
Patient-specific TAVR model
FEA simulation — Case 1
FEA simulation — Case 2
Cardio4D uses Finite Element Analysis (FEA) to virtually deploy a valve and measure the resulting wall tension. If the predicted stress exceeds the tissue's failure threshold, the software flags a "High Risk of Rupture" warning to the clinician. By accurately modeling the stiffness of calcium deposits, our software shows how a rigid calcified nodule will be pushed into the aortic wall during expansion — a key predictor of rupture that cannot be seen on static 2D images. [Wang et al., 2015] [Wang et al., 2014]
Coronary Obstruction Analysis
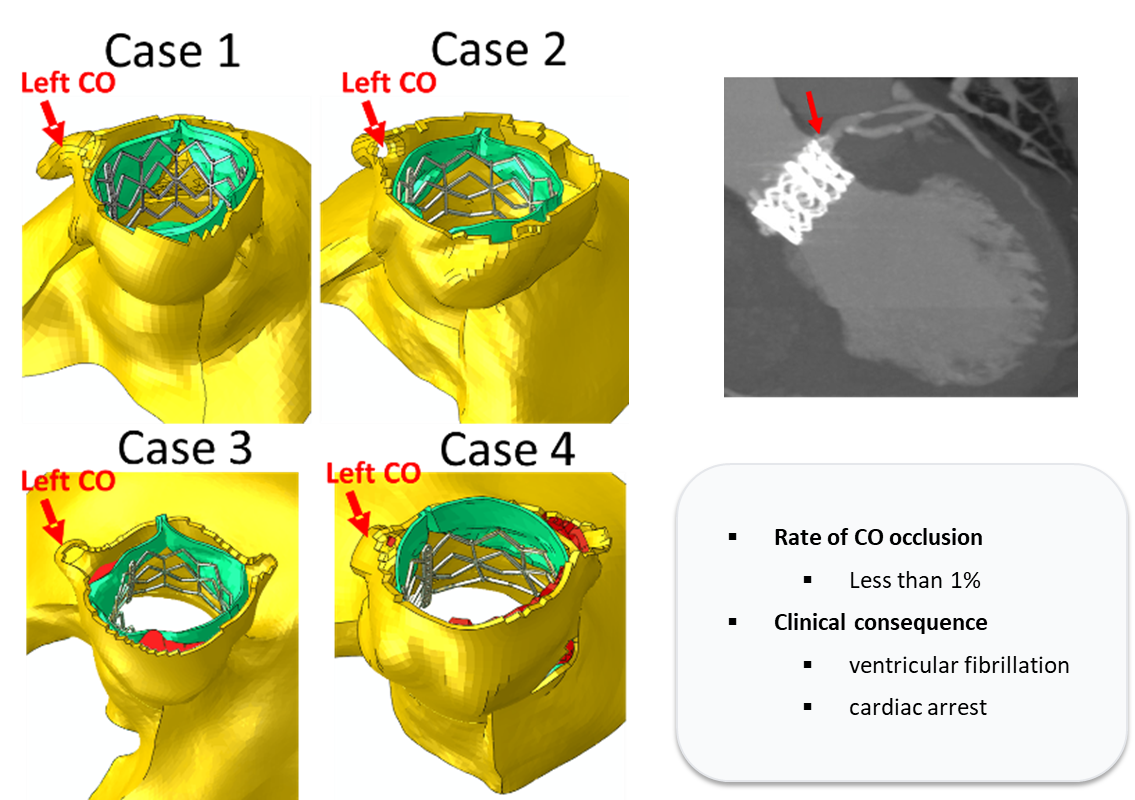
Coronary ostia displacement analysis
Coronary Obstruction (CO) is a severe adverse event in TAVR, occurring in less than 1% of cases but carrying a high mortality rate. Our platform utilizes FEA-based TAVR deployment simulations to visualize the mechanical displacement of native leaflets toward the coronary ostia, allowing clinicians to preemptively detect and avoid potential occlusion. [Wang et al., 2015]
Paravalvular Leakage & Sizing Optimization
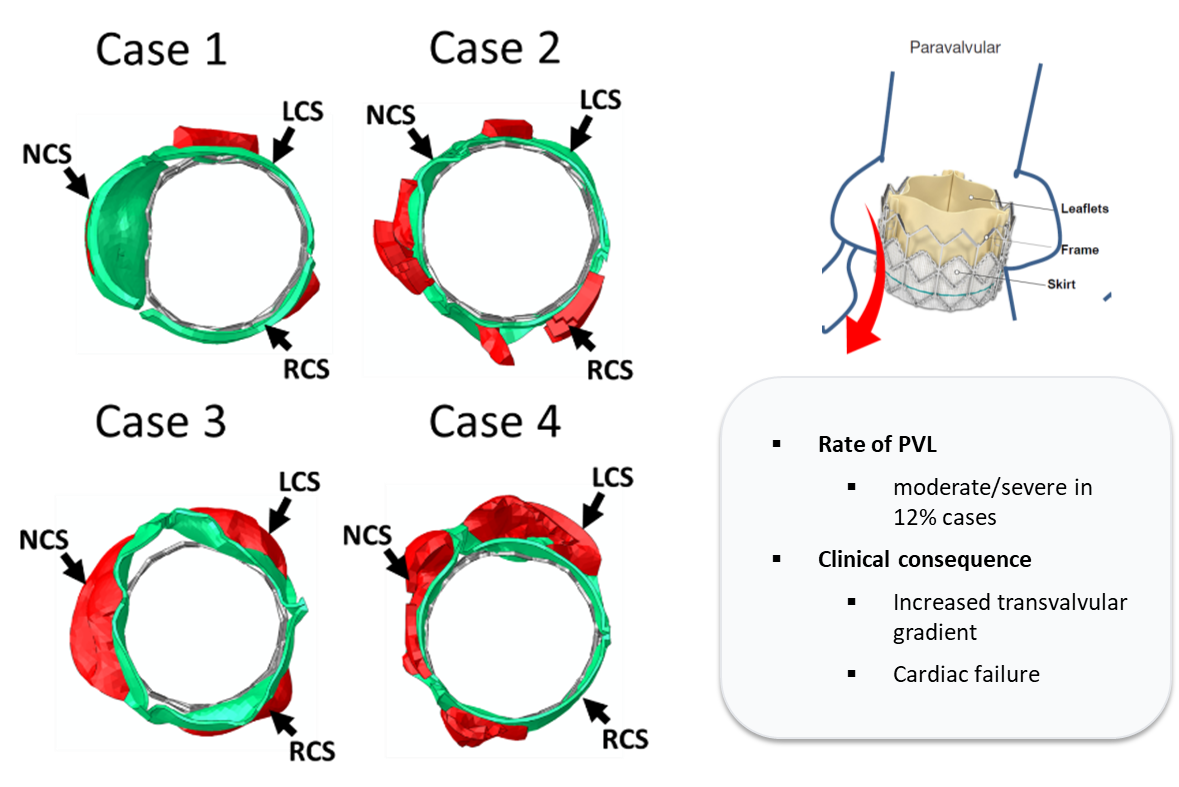
Paravalvular leak assessment
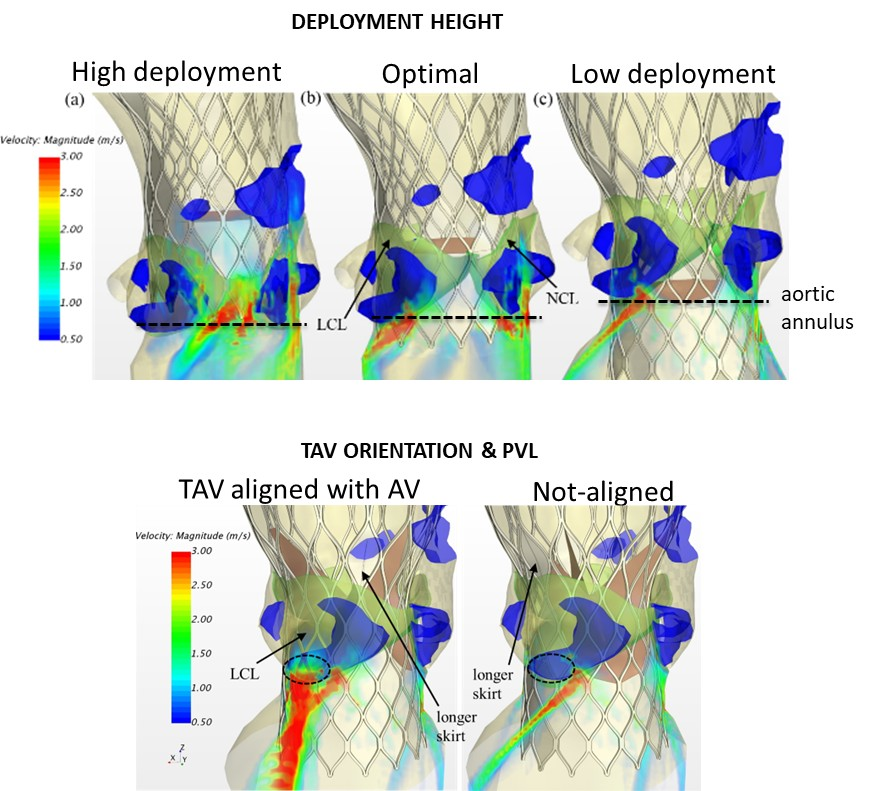
Deployment height optimization
Paravalvular Leakage (PVL) remains a critical complication in TAVR, leading to increased transvalvular gradients and heart failure. While moderate to severe PVL occurs in approximately 12% of cases, research by Généreux et al. (2013) established that even mild PVL significantly correlates with increased late mortality. By identifying annular calcification and valve undersizing as the primary drivers of leakage, this research directly supports the clinical necessity for high-precision, patient-specific planning software to optimize device selection and placement. Cardio4D's algorithms recommend the optimal valve size or deployment strategy to achieve effective valve function while staying below the mechanical "danger zone" for each patient's anatomy. [Sun et al., 2010]
Impact on Mitral Valve Function
Bird's-eye view — MV interaction
Side-elevation view — MV interaction
By transforming static patient imaging into a dynamic, fully-coupled mechanical model, the platform allows clinicians to virtually test different deployment heights — High, Optimal, and Low — before the procedure begins. This enables precise prediction of how a TAVR stent's position will mechanically compress the aortic-mitral curtain and influence concomitant mitral regurgitation. Cardio4D provides a quantitative "sweet spot" for implantation, ensuring that aortic repair does not compromise mitral valve function. [Mao et al., 2017]
Fluid-Structure Interaction (FSI)
Fully-coupled hemodynamic simulation of the left heart
MV & LV blood flow simulation
TAVR hemodynamic flow — View 1
TAVR hemodynamic flow — View 2
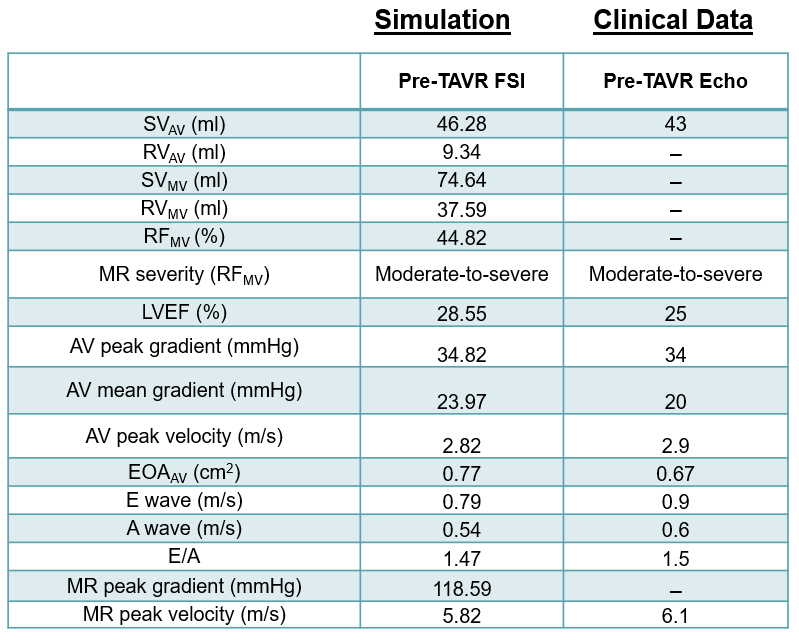
Hemodynamic analysis data
Cardio4D represents a major leap in computational cardiology by being the first to simulate the fully-coupled interaction between the aortic and mitral valves within a realistic, patient-specific 3D left ventricle model. Unlike previous research that looked at valves in isolation, this work uses Fluid-Structure Interaction (FSI) to capture how blood flow and tissue deformation influence one another simultaneously. The simulation accurately maps high-velocity regurgitant jets and hemodynamic patterns across the entire left heart apparatus, providing a holistic view of cardiac function during the full heart cycle. [Mao et al., 2017] [Qin et al., 2024]
Transcatheter Mitral Valve Replacement (TMVR)
Edge-to-Edge Repair optimization through computational biomechanics
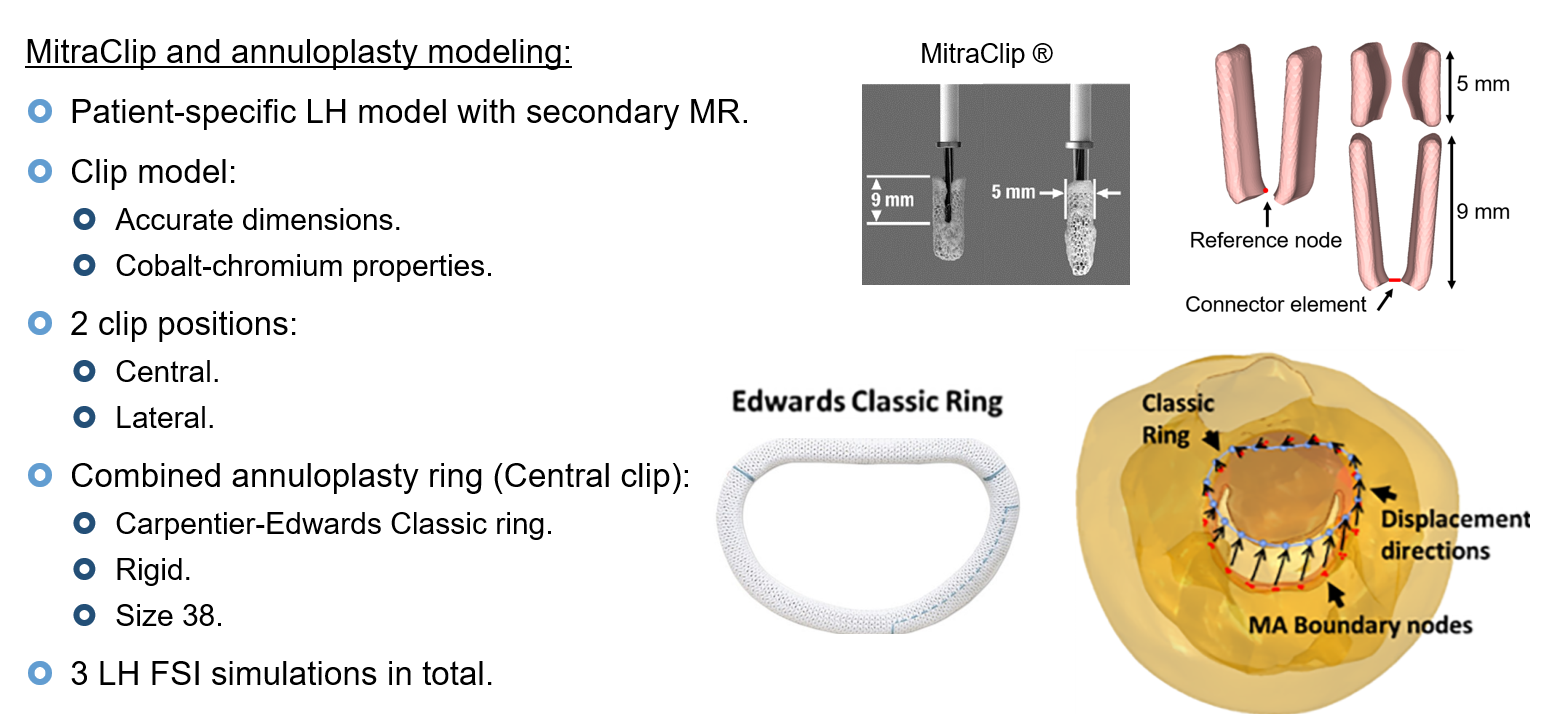
Clip & ring stress analysis — overview
TEER clip deployment simulation
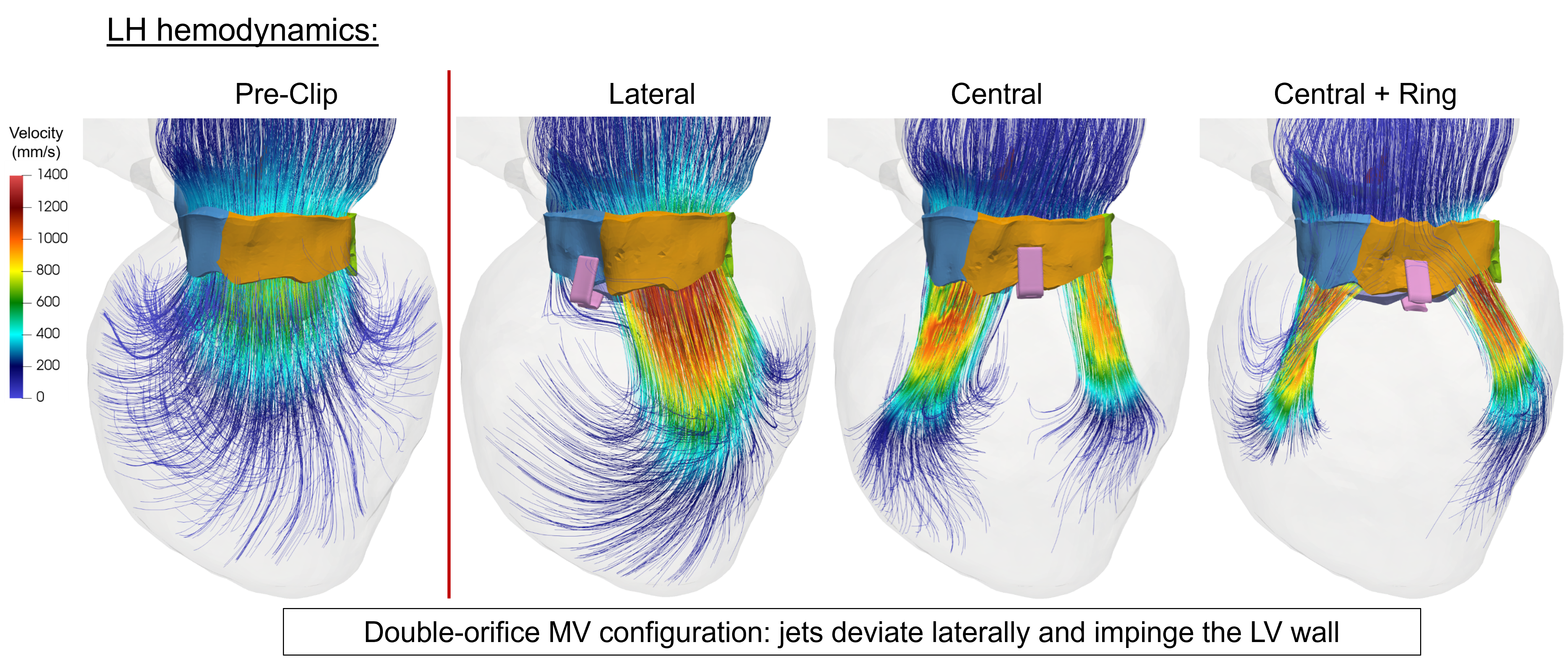
Flow — lateral orifice
Flow — central orifice
Flow — central ring configuration
Leaflet stress distribution
Cardio4D utilizes patient-specific Finite Element Analysis to quantify the biomechanical impact of Transcatheter Edge-to-Edge Repair (TEER) on the mitral valve. By simulating the creation of a "double-orifice" geometry, the simulation result demonstrates how the procedure fundamentally redistributes mechanical stress across the leaflets and chordae tendineae. Cardio4D leverages these findings to allow clinicians to virtually test clip placements, optimizing for both reduced regurgitation and long-term structural durability. The platform's algorithms accurately predict "hot spots" of high stress at the clip-leaflet interface, helping to identify and mitigate risks of tissue tearing or device detachment. Ultimately, this work transforms TEER from a reactive intervention into a data-driven, predictive procedure tailored to each patient's unique anatomy. [Mao et al., 2020] [Caballero et al., 2018]
Artificial Intelligence (AI) and Deep Learning
Real-time biomechanical prediction through deep learning surrogates
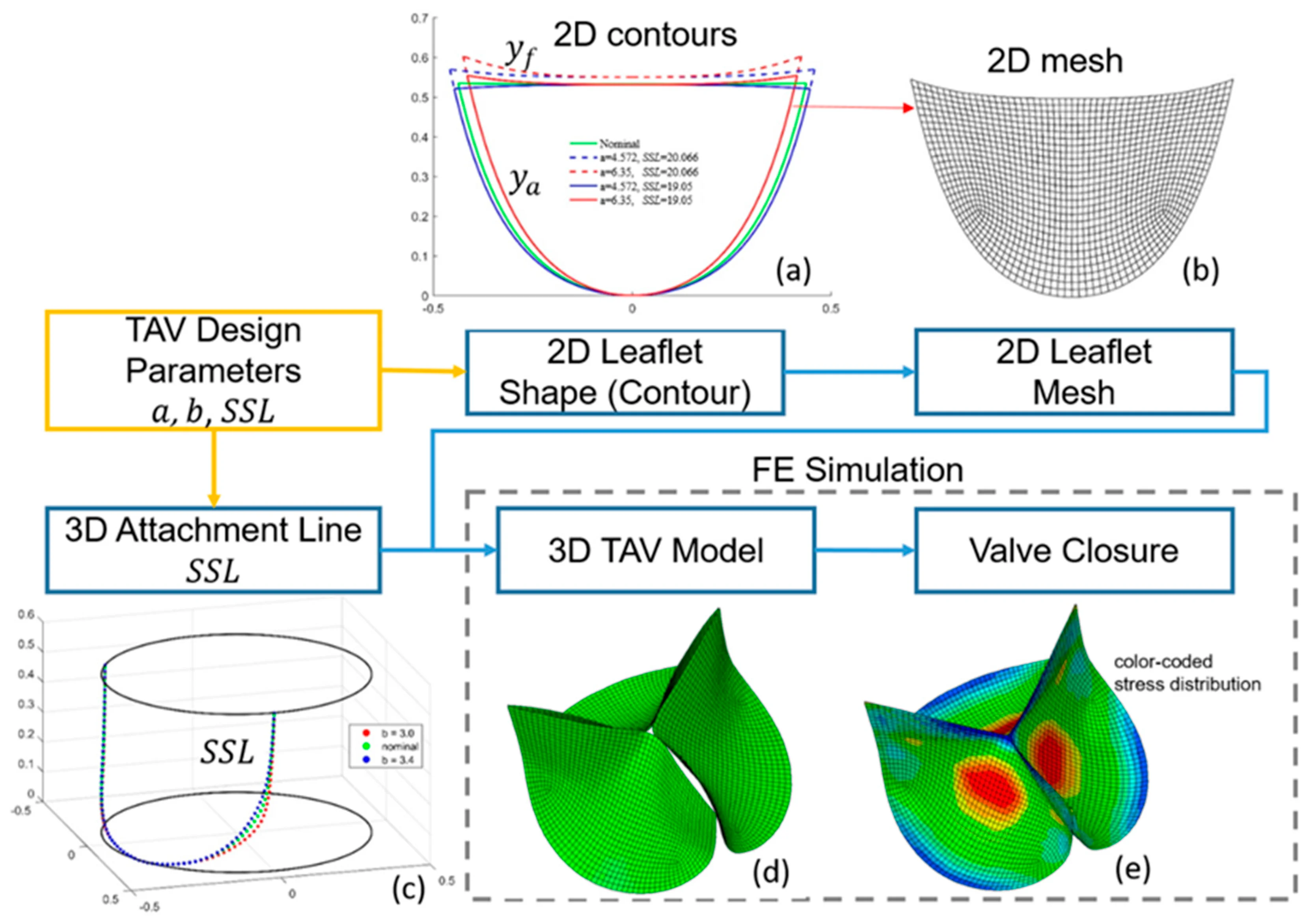
Machine learning for valve leaflet design
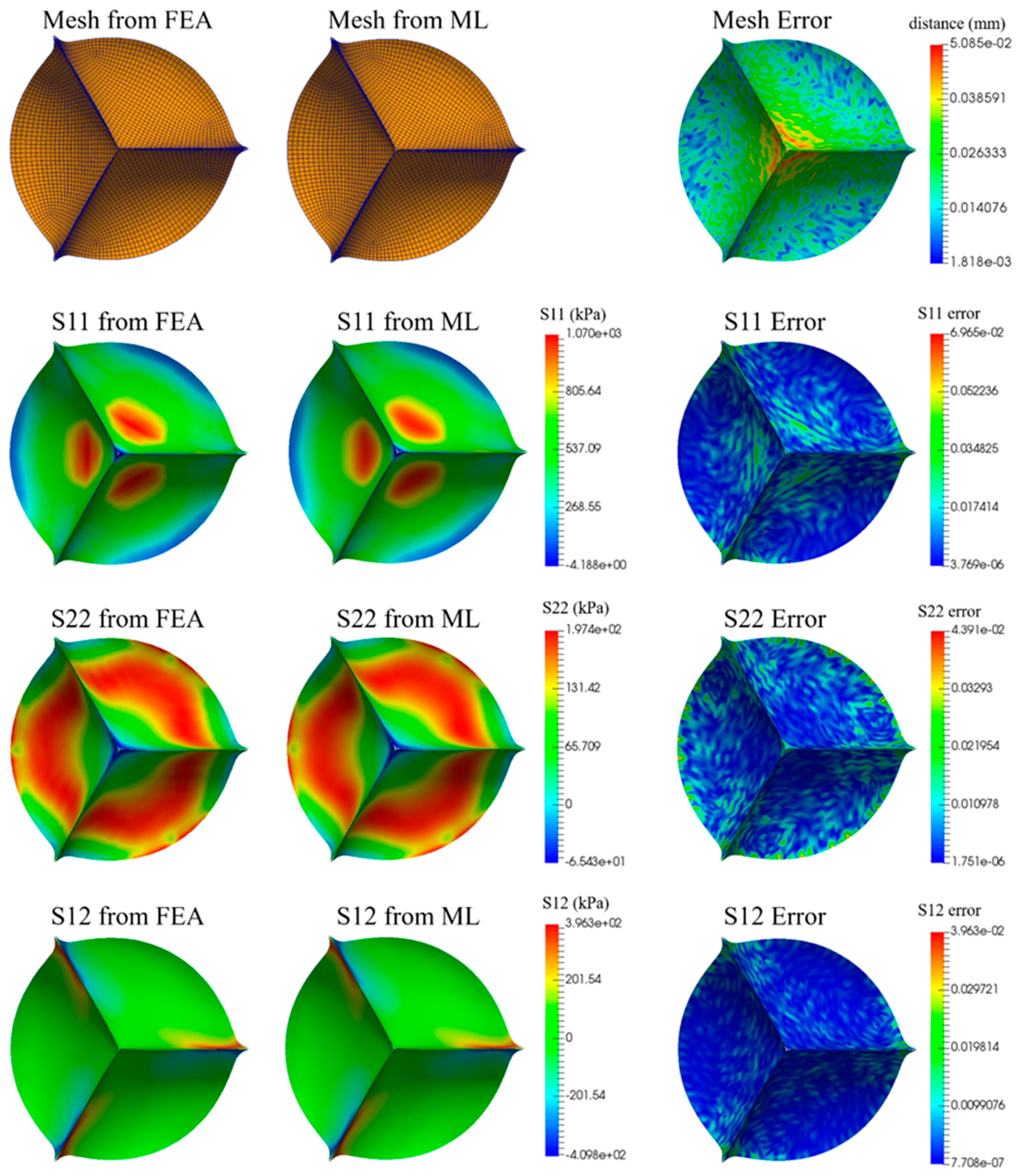
ML-predicted valve stress analysis
A proof of concept for using machine learning to revolutionize the design and analysis of artificial heart valves, specifically by predicting mechanical stress based on leaflet geometry. By replacing traditional, computationally expensive simulations with deep learning surrogates, we demonstrate that high-fidelity biomechanical performance can be evaluated in milliseconds with remarkable accuracy. [Liang et al., 2020]
Summary
The Cardio4D team has established a rigorous engineering foundation by combining high-fidelity Fluid-Structure Interaction (FSI) with patient-specific anatomical modeling to predict complex heart valve behavior. By quantifying the mechanical impact of fiber orientation, tissue anisotropy, and device eccentricity, these computational models provide the mathematical frameworks necessary to transform static clinical images into dynamic, functional simulations. The model prediction specifically addresses critical procedural risks — such as paravalvular leak, aortic root rupture, and coronary occlusion — by identifying the biomechanical "danger zones" unique to each patient's calcification patterns. Furthermore, the integration of Deep Learning surrogates and automated 3D reconstruction allows for these complex calculations to be performed with the speed required for real-time clinical decision-making.
Clinical Confidence
By using validated models, Cardio4D offers clinicians a "virtual dry run" that has been peer-reviewed in top journals, reducing the uncertainty of complex interventions.
Precision Targeting
The platform identifies the "Optimal" deployment height to mitigate concomitant mitral regurgitation — a level of precision that standard imaging cannot provide.
Safety Benchmarking
The software uses established mechanical failure thresholds to flag risks like aortic rupture before a balloon is ever expanded, turning "black swan" events into predictable data points.
Reduced Complications
By optimizing for eccentricity and effective orifice area, Cardio4D ensures that devices don't just "fit," but function harmoniously with the patient's unique hemodynamics to prevent premature fatigue.
References
- Mao, W., Caballero, A., & Sun, W. (2020). Biomechanical impact of transcatheter edge-to-edge repair on the mitral valve: A patient-specific computational study. Journal of Biomechanics, 106, 109823.
- Mao, X., Wang, Q., Sun, W., & Sacks, M. S. (2018). Influence of leaflet fiber orientation on the mechanical environment of the aortic valve. Journal of Biomechanical Engineering, 140(2), 021010.
- Caballero, A., Mao, W., McKay, R., Primiano, C., Hashim, S., & Sun, W. (2018). New insights into mitral heart valve prolapse after chordae rupture through fluid-structure interaction computational modeling. Scientific Reports, 8(1), 17306.
- Pham, T., Sun, W., Fan, R., & He, Z. (2017). Finite Element Analysis of Patient-Specific Mitral Valve with Mitral Regurgitation. Cardiovascular Engineering and Technology, 8(1), 3-16.
- Mao, W., Caballero, A., McKay, R., Primiano, C., & Sun, W. (2017). Fully-coupled fluid-structure interaction simulation of the aortic and mitral valves in a realistic 3D left ventricle model. PLOS ONE, 12(9), e0184729.
- Mao, W., & Sun, W. (2015). Computational modeling of the mitral valve: A review. Computers in Biology and Medicine, 60, 26-32.
- Qin, T., Mao, W., Caballero, A., et al. (2024). Patient-specific analysis of bicuspid aortic valve hemodynamics using a fully coupled fluid-structure interaction model. Computers in Biology and Medicine, 172, 108191.
- Wang, Q., Kodali, S., Primiano, C., & Sun, W. (2015). Simulations of transcatheter aortic valve implantation: implications for aortic root rupture. Biomechanics and Modeling in Mechanobiology, 15(1), 29-43.
- Wang, Q., Sirois, E., & Sun, W. (2014). Patient-specific modeling of transcatheter aortic valve implantation. Journal of Cardiovascular Translational Research, 7(2), 214-227.
- Wang, Q., Sun, W., & Sacks, M. S. (2014). A structural constitutive model of the aortic valve leaflet. Biomechanics and Modeling in Mechanobiology, 13(6), 1335-1349.
- Sun, W., Li, K., & Sirois, E. (2010). Simulated elliptical bioprosthetic valve deformation: implications for asymmetric transcatheter valve deployment. Journal of Biomechanics, 43(16), 3085-3090.
- Fan, R., Sun, W., & Sacks, M. S. (2013). Modeling the biaxial mechanical behavior of heart valve tissues. Journal of Biomechanical Engineering, 135(2), 021008.
- Sacks, M. S., & Sun, W. (2003). Multiaxial mechanical behavior of biological materials. Annual Review of Biomedical Engineering, 5(1), 251-284.
- Sun, W., & Sacks, M. S. (2005). Finite element implementation of a structural constitutive model for planar soft tissues. Computer Methods in Biomechanics and Biomedical Engineering, 8(1), 33-39.
- Kong, F., Pham, T., Martin, C., et al. (2018). Finite element analysis of annuloplasty and papillary muscle relocation on a patient-specific mitral regurgitation model. PLOS ONE, 13(6), e0198331.
- Martin, C., & Sun, W. (2020). A deep learning approach to estimate stress distribution: a fast and accurate surrogate of finite-element analysis. Journal of The Royal Society Interface.
- Généreux, P., Head, S. J., Hahn, R. T., et al. (2013). Paravalvular leak after transcatheter aortic valve replacement: the new Achilles' heel? Journal of the American College of Cardiology, 61(11), 1125-1136.
- Pham, T., et al. (2017). Quantification and comparison of the mechanical properties of four human cardiac valves. Acta Biomaterialia.