Thoracic Aortic Aneurysm (TAA) Rupture Risk Analysis
Our foundation is built on a deep pedigree of research published in leading medical and engineering journals. We bridge the gap between engineering and medicine to solve the most complex challenges in structural heart disease through advanced constitutive modeling, patient-specific simulation, and structural biomechanics.
Aneurysm Risk & Artificial Intelligence (AI)
Patient-specific biomechanical risk assessment beyond diameter
Prevalence & The Aging Population: While Abdominal Aortic Aneurysms (AAA) affect 6-9% of adults over 65, TAA represents a more specialized and lethal threat. Its incidence peaks sharply between ages 65 and 70, affecting up to 0.5% of the general population — many of whom remain undiagnosed. [Wanhainen et al., 2019] [Olsson et al., 2006]
The Silent Threat: TAA is notoriously asymptomatic, often remaining undetected until a catastrophic rupture or dissection occurs. This "silent" progression makes it a Top 20 cause of death in the United States. [Elefteriades & Farkas, 2010]
The 5.5 cm Failure: Current clinical guidelines rely almost exclusively on a 5.5 cm diameter threshold for surgical intervention. However, research shows that a significant percentage of patients experience life-threatening dissections or ruptures below this threshold. [Pape et al., 2007]
The Cardio4D Solution: Relying on diameter alone is insufficient because it ignores the underlying physics of the aortic wall. Cardio4D's biomechanical risk modeling fills this unmet need by quantifying peak wall stress, providing a precise, patient-specific metric that identifies high-risk patients who would otherwise be missed by traditional measurements.
Aortic Wall Stress Analysis
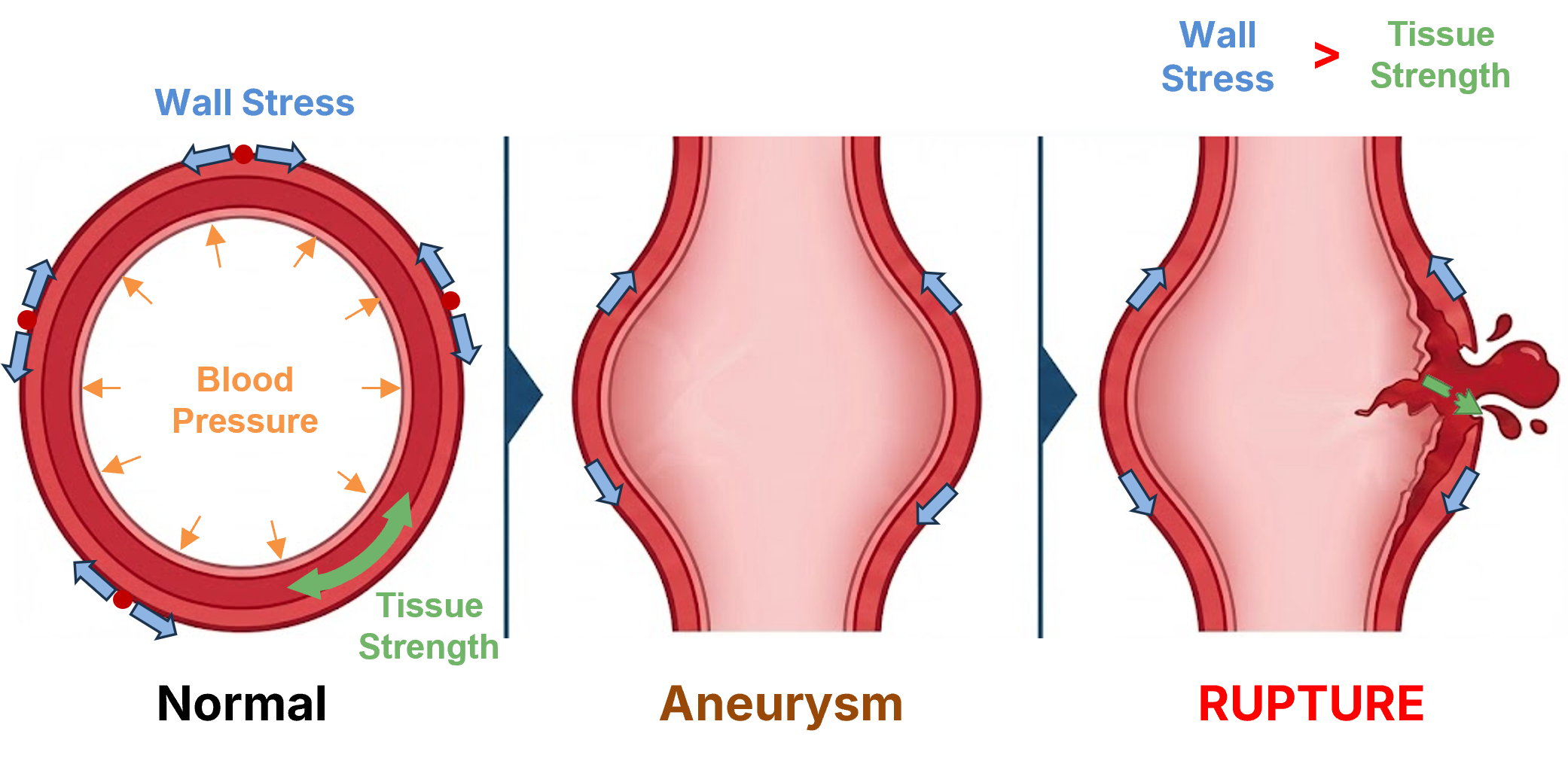
Aortic wall stress distribution — insight into rupture risk beyond diameter
Aortic wall stress distribution provides insight into rupture risk beyond diameter. The rupture/dissection mechanism occurs when wall stress exceeds tissue strength. Our patient-specific risk assessment incorporates geometry and biomechanics.
Validated Predictive Power
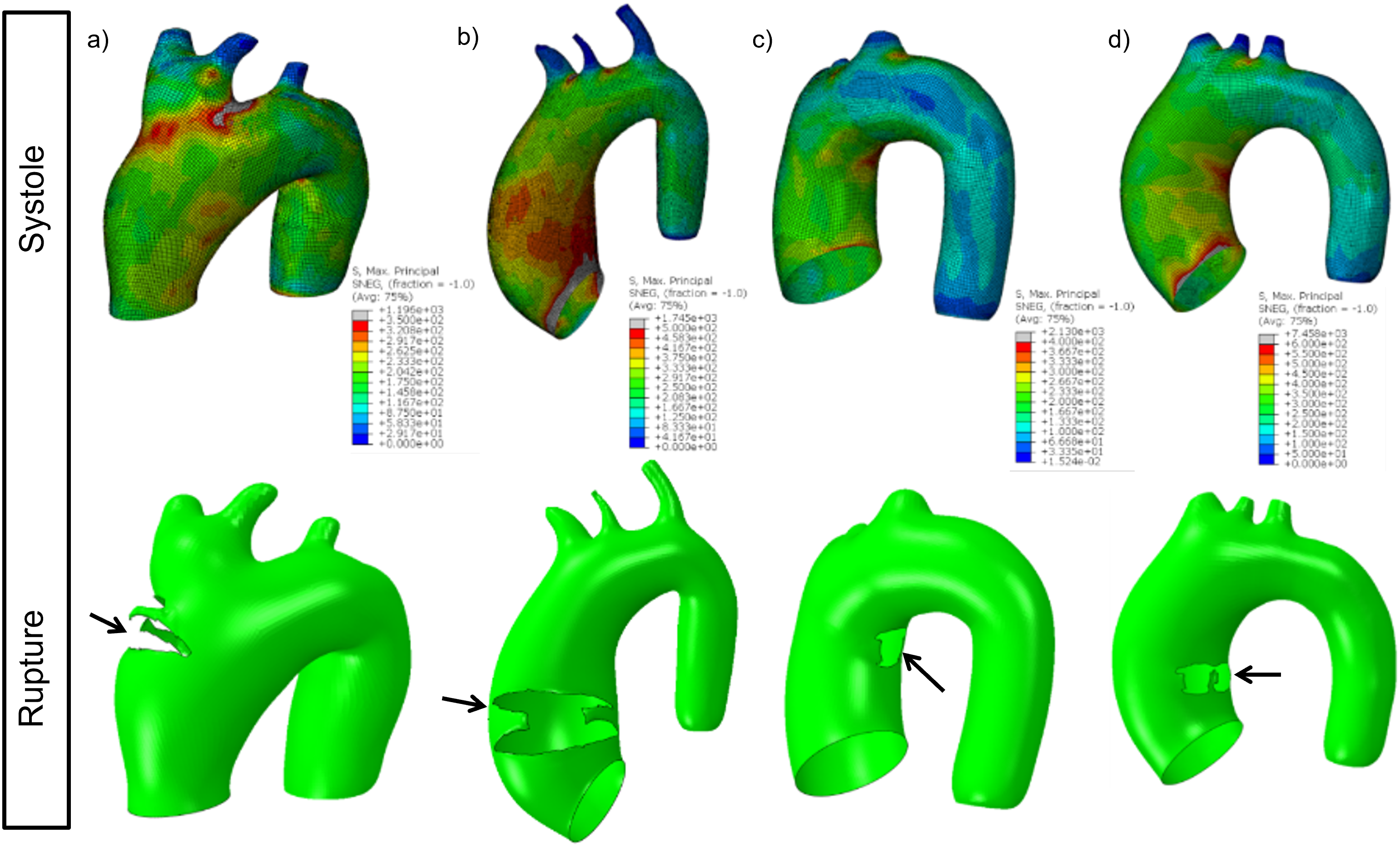
Patient-specific ascending aortic aneurysm analysis
Cardio4D delivers a risk assessment engine grounded in clinical validation. By using the peak wall stress (PWS) thresholds established in peer-reviewed studies, the platform can flag "high-stress/small-diameter" patients who are currently missed by standard care guidelines. We provide the methodology for transforming patient-specific CT data into high-fidelity meshes, ensuring that the 3D models used for surgical planning are not just visualizations, but accurate biomechanical twins of the patient's heart. [Martin, Sun & Elefteriades, 2015] [Pham et al., 2013]
Machine Learning Surrogate for FEA
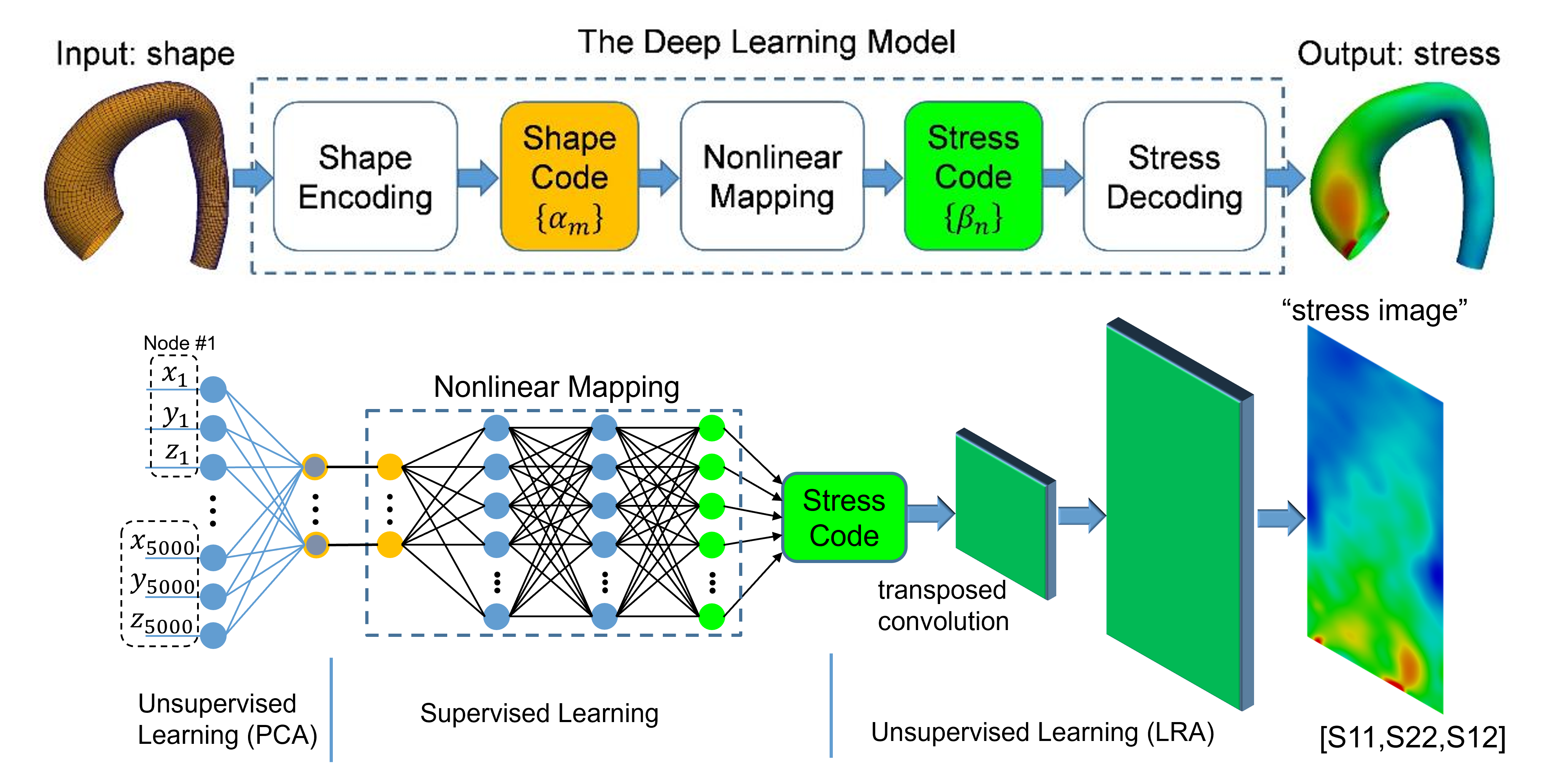
Deep learning surrogate for wall stress prediction
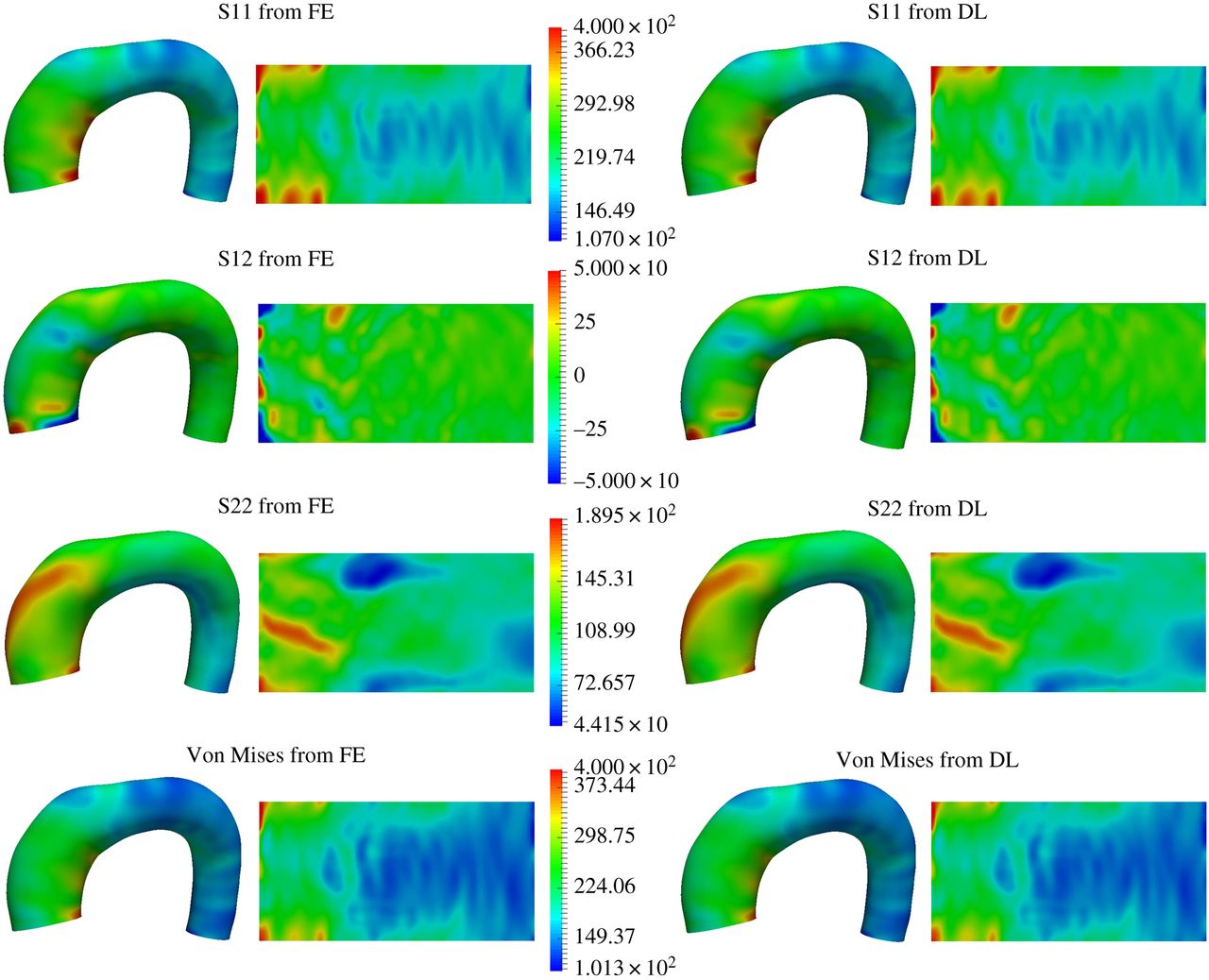
ML vs FEA accuracy comparison — Case 1
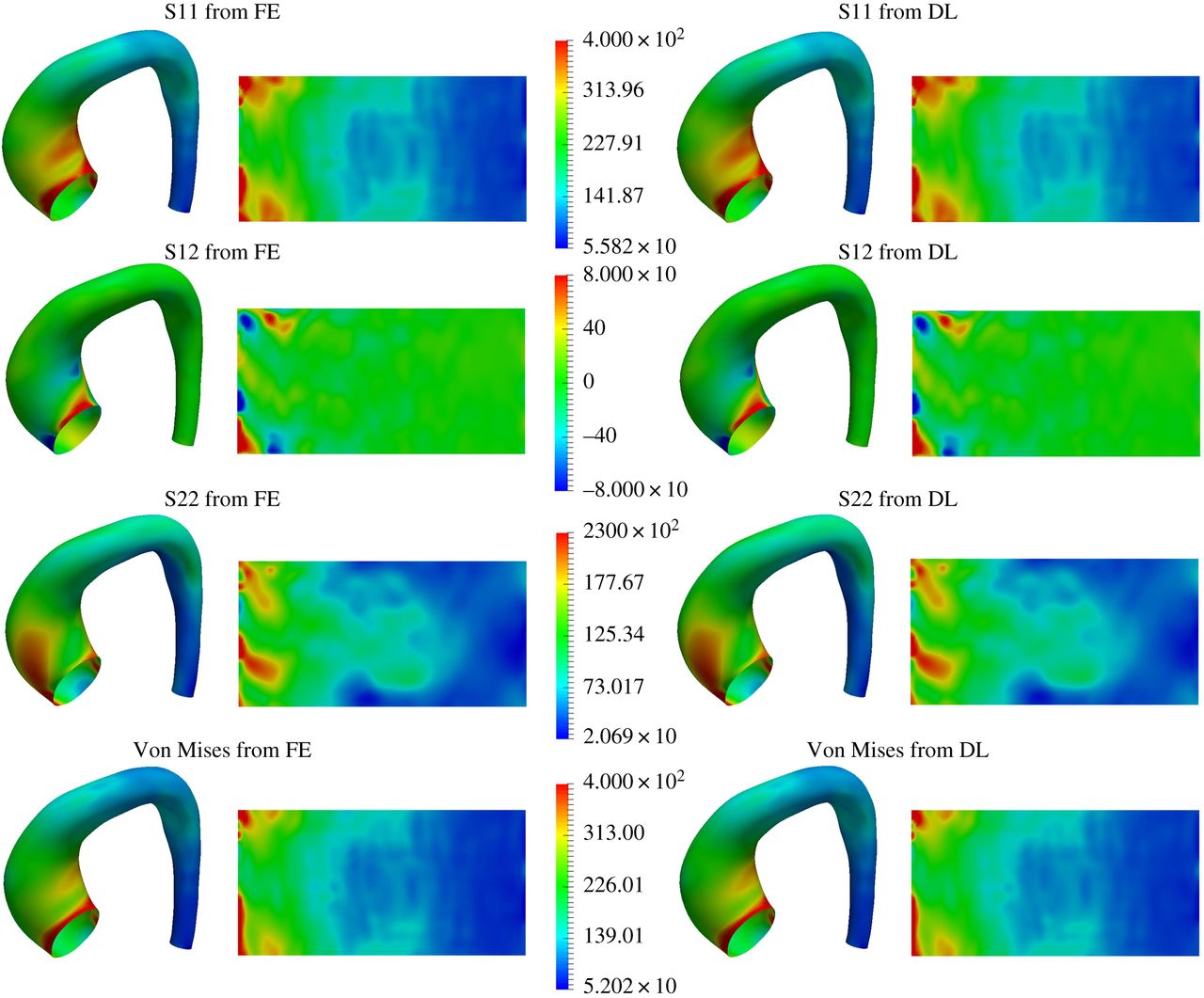
ML vs FEA accuracy comparison — Case 2
It has been rigorously shown that advanced deep learning models can successfully replace traditional, time-consuming engineering simulations with near-instantaneous predictions. By achieving over 95% accuracy in estimating complex arterial wall stress compared to gold-standard Finite Element Analysis, Cardio4D establishes the scientific proof of concept for a real-time diagnostic engine. These findings confirm that high-fidelity biomechanical insights can be delivered at clinical speeds without compromising the mathematical precision required for life-critical surgical planning. [Liang et al., 2018]
AI-Powered Biomechanical Analysis

Pre/post stress analysis — Case 1
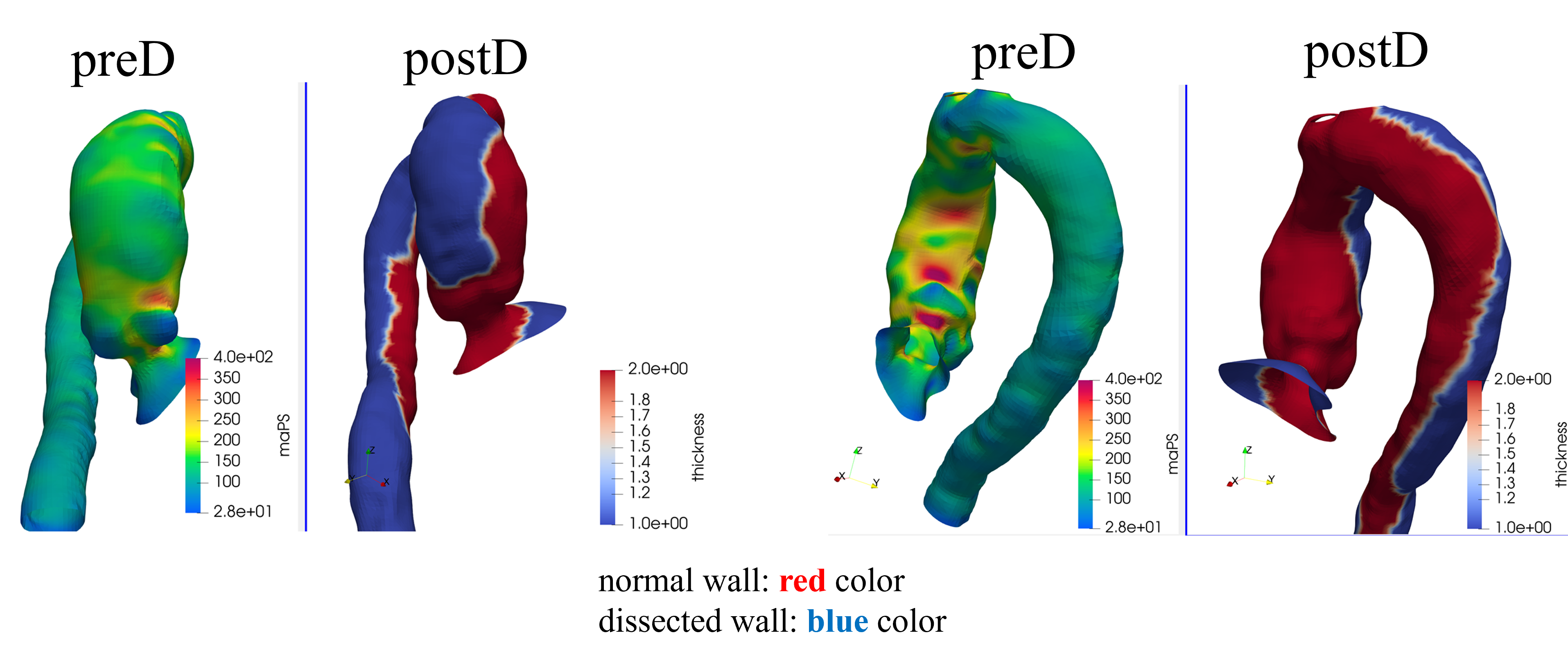
Pre/post stress analysis — Case 2
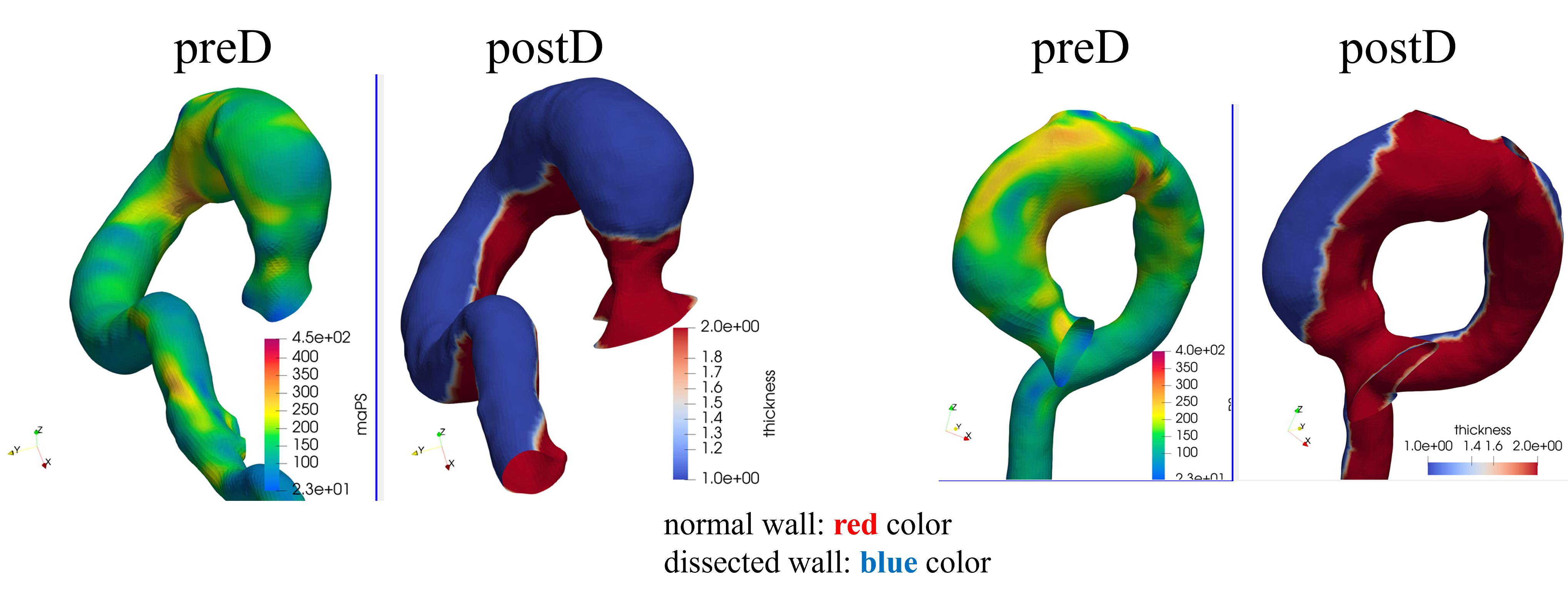
Pre/post stress analysis — Case 3
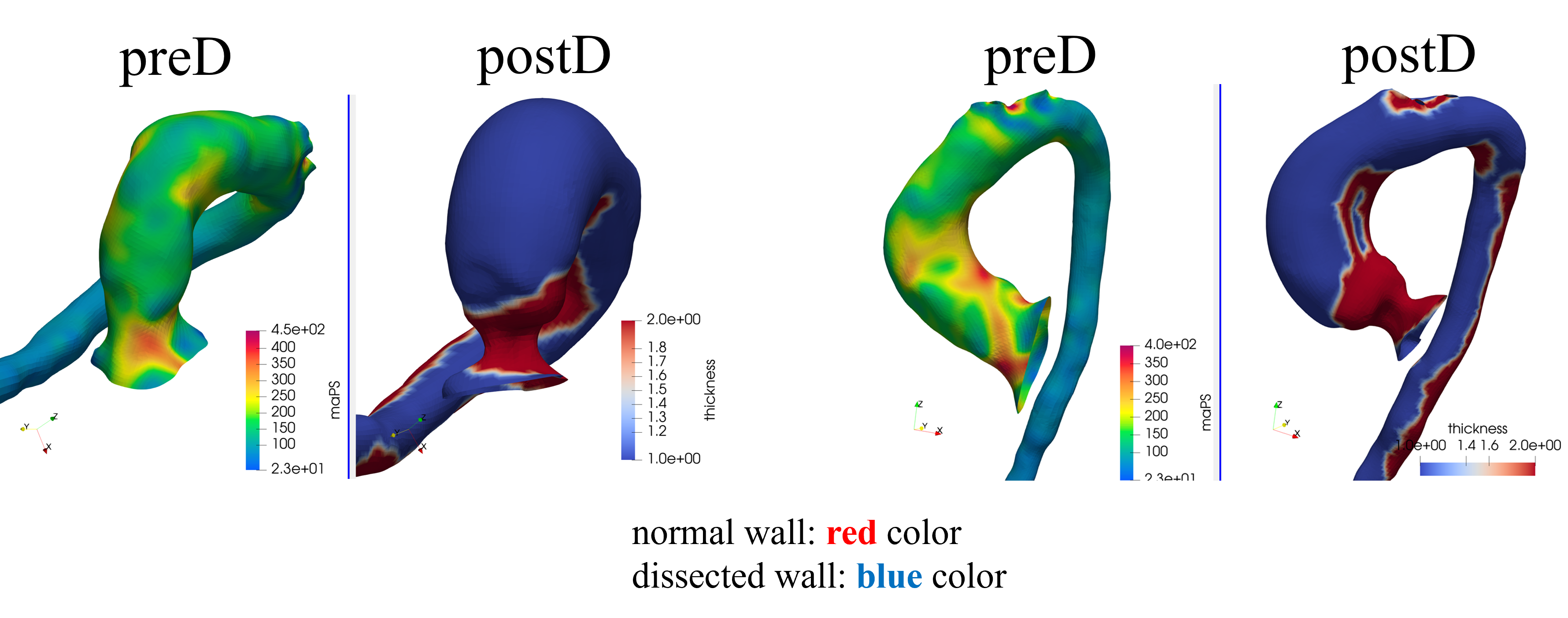
Pre/post stress analysis — Case 4
Cardio4D leverages PyTorch-FEA (DNN-FEA integration), a breakthrough framework that integrates traditional finite element analysis with automatic differentiation to enable high-speed, gradient-based optimization of biomechanical models. By utilizing the Autograd engine, the platform performs seamless, real-time estimation of material parameters and wall stress distributions directly from clinical imaging data. Unlike traditional solvers that require hours of manual iteration, Cardio4D's AI-powered inverse modeling engine "back-calculates" a patient's specific aortic tissue stiffness in seconds, providing a truly personalized rupture risk assessment. [Liang et al., 2018] [Liang et al., 2023]
Summary
Cardio4D: Redefining Cardiovascular Precision
Cardio4D provides the high-performance computational engine that replaces slow, manual engineering with automated, AI-driven workflows. By fusing Artificial Intelligence with Finite Element Analysis (FEA), we deliver detailed biomechanical risk reports in near real-time, facilitating immediate point-of-care decisions when every second counts.
Beyond Diameter
The implementation of Statistical Shape Modeling (SSM) and our proprietary PyTorch-FEA integration enables the platform to move beyond antiquated, diameter-based measurements. Cardio4D provides a high-fidelity classification of rupture risk based on the patient's unique, four-dimensional aortic geometry and in-vivo tissue stiffness.
Research to Reality
This deep pedigree of validated research transforms Cardio4D from a rigorous academic tool into a scalable, AI-powered diagnostic platform. We are not just visualizing anatomy; we are quantifying the physics of human life — redefining the standard of cardiovascular care and bringing world-class biomechanical insight to every surgical suite.
References
- Liang, L., Liu, M., Elefteriades, J., & Sun, W. (2023). PyTorch-FEA: Autograd-enabled finite element analysis methods with applications for biomechanical analysis of human aorta. Computer Methods and Programs in Biomedicine, 240, 107715.
- Liang, L., Liu, M., Martin, C., & Sun, W. (2018). A deep learning approach to estimate stress distribution: a fast and accurate surrogate of finite-element analysis. Journal of The Royal Society Interface, 15(138), 20170844.
- Liang, L., et al. (2020). A proof of concept study of using machine-learning in artificial aortic valve design: From leaflet design to stress analysis. Bioengineering, 6(4), 104.
- Liang, L., Mao, W., & Sun, W. (2020). A feasibility study of deep learning for predicting hemodynamics of human thoracic aorta. Journal of Biomechanics, 99, 109544.
- Liang, L., Liu, M., Martin, C., & Sun, W. (2018). A machine learning substitution of finite element analysis for quantifying arterial wall stress. Computer Methods in Applied Mechanics and Engineering, 338, 209-223.
- Liang, L., Kong, F., Martin, C., Pham, T., Wang, Q., Duncan, J. S., & Sun, W. (2016). Machine learning-based 3-D geometry reconstruction and modeling of aortic valve deformation using 3-D computed tomography images. International Journal for Numerical Methods in Biomedical Engineering, 32(5), e2735.
- Liang, L., Liu, M., Martin, C., Elefteriades, J. A., & Sun, W. (2017). A machine learning approach to investigate the relationship between shape features and numerically predicted risk of ascending aortic aneurysm. Biomechanics and Modeling in Mechanobiology, 16(5), 1519-1533.
- Liang, L., Liu, M., Martin, C., & Sun, W. (2018). A machine learning approach as a surrogate of finite element analysis-based inverse method to estimate the zero-pressure geometry of human thoracic aorta. International Journal for Numerical Methods in Biomedical Engineering, 34(8).
- Liu, M., Liang, L., & Sun, W. (2019). Estimation of in vivo constitutive parameters of the aortic wall: a machine learning approach. Computer Methods in Applied Mechanics and Engineering.
- Pham, T., Martin, C., Elefteriades, J., & Sun, W. (2013). Biomechanical characterization of ascending aortic aneurysm with concomitant bicuspid aortic valve and bovine aortic arch. Acta Biomaterialia, 9(8), 7927-7936.
- Martin, C., Sun, W., & Elefteriades, J. (2015). Patient-specific finite element analysis of ascending aorta aneurysms. American Journal of Physiology-Heart and Circulatory Physiology, 308(10), H1306-H1316.
- Martin, C., Pham, T., & Sun, W. (2013). Biomechanical Characterization of Human Ascending Aortic Aneurysm Tissues. Cardiovascular Engineering and Technology, 4(4), 385-399.
- Martin, C., et al. (2017). Morphometric Analysis of the Ascending Aorta: Implications for Aortic Aneurysm Repair. Journal of Biomechanics.
- Qin, T., et al. (2024). Patient-specific analysis of bicuspid aortic valve (BAV) hemodynamics using a fully coupled fluid-structure interaction model. Computers in Biology and Medicine.
- Pak, D. H., Liu, M., ... Liang, L., ... & Sun, W. (2024). Patient-specific Heart Geometry Modeling for Solid Biomechanics using Deep Learning. PMC10764002.
- Liang, L., et al. (2016). Estimation of aortic valve leaflets from 3D CT images using local shape dictionaries and linear coding. SPIE Medical Imaging.
- Liang, L., et al. (2017). A Machine Learning-Based Method for Quantifying Wall Stress in Ascending Aortic Aneurysms. Journal of Biomechanical Engineering.
- Martin, C., & Sun, W. (2015). Biomechanical Characterization of Bovine and Porcine Pericardium for Transcatheter Heart Valve Biomaterials. Cardiovascular Engineering and Technology.
- Wanhainen, A., et al. (2019). ESVS 2019 Clinical Practice Guidelines on the Management of Abdominal Aorto-iliac Artery Aneurysms. European Journal of Vascular and Endovascular Surgery.
- Elefteriades, J. A., & Farkas, E. A. (2010). Thoracic Aortic Aneurysm: Clinically Pertinent Fundamentals. Journal of the American College of Cardiology.
- Olsson, C., et al. (2006). Thoracic aortic aneurysm and dissection: increasing incidence and improved outcomes. Circulation, 114(24), 2611-2618.
- Pape, L. A., et al. (2007). Presentation, Diagnosis, and Outcomes of Acute Aortic Dissection: 17-Year Trends from IRAD. Journal of the American College of Cardiology.
- Clouse, W. D., et al. (1998). The continuity of medical and surgical management of thoracic aortic aneurysms. Mayo Clinic Proceedings, 73(12), 1132-1137.
- Kuzmik, G. A., et al. (2012). Natural history of thoracic aortic aneurysms. Journal of Vascular Surgery, 56(2), 565-571.